Main Article Content
Abstract
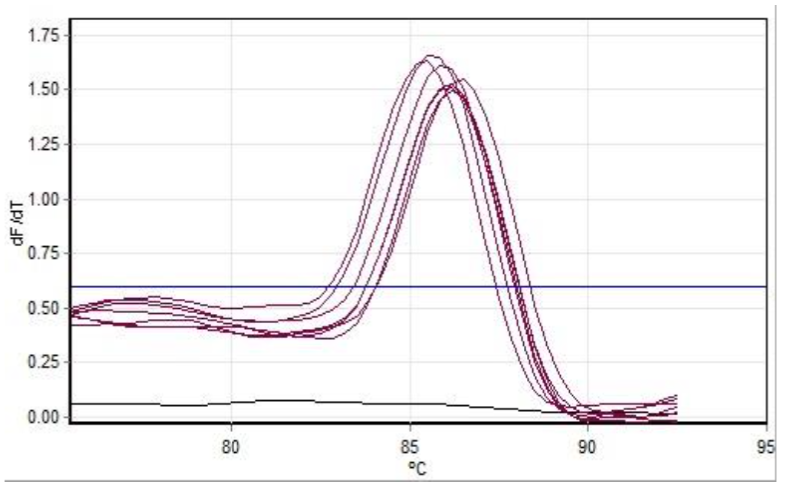
Detection of Salmonella typhimurium ATCC 14028 bacteria in bakery product samples by real-time PCR using boiling isolation technique. The basis of this research is to have an impact on economic value in carrying out the confirmation test for Salmonella typhimurium ATCC 14028, where testing is carried out conventionally will require large costs, so it is necessary to innovate in terms of modifying the testing phase so that it is more effective and efficient. The purpose of this study was to see whether the boiling isolation technique could be used for the detection test for Salmonella typhimurium ATCC 14028 on bacterial product samples. The sample in this study consisted of 15 types of bacterial product samples spiked with Salmonella typhimurium ATCC 14028 cultures that had been cultured into phase 2 working cultures. The method used in this study was qPCR analysis using the SYBR Green method. The results of real-time PCR analysis obtained Ct values in the range 7.55 - 8.91 with an average of 8.28 and a Tm value in the range 85.50 - 86.20 with an average of 85.77 Based on these data it can be concluded that the detection of Salmonella typhimurium bacteria ATCC 14028 with real-time PCR using boiling isolation technique can be applied for testing on bakery product samples.
Keywords
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- Ahmed, O. B., & Dablool, A. S. (2017). Quality improvement of the DNA extracted by boiling method in gram negative bacteria. International Journal of Bioassays, 6(4), 5347–5349.
- Ahsani, M. R., & Shamsaddini, B. M. (2013). Compare of two methods of direct PCR and PCR with DNA extraction in Clostridium perfringens typing.
- Alves, J., Hirooka, E. Y., & de Oliveira, T. C. R. M. (2016). Development of a multiplex real-time PCR assay with an internal amplification control for the detection of Campylobacter spp. and Salmonella spp. in chicken meat. LWT-Food Science and Technology, 72, 175–181.
- Dimitrakopoulou, M.-E., Stavrou, V., Kotsalou, C., & Vantarakis, A. (2020). Boiling extraction method VS commercial Kits for bacterial DNA isolation from food samples. Journal of Food Science and Nutrition Research, 3(4), 311–319.
- Giacomazzi, S., Leroi, F., & Joffraud, J. (2005). Comparison of three methods of DNA extraction from cold‐smoked salmon and impact of physical treatments. Journal of Applied Microbiology, 98(5), 1230–1238.
- Guerra, B., Junker, E., Schroeter, A., Malorny, B., Lehmann, S., & Helmuth, R. (2003). Phenotypic and genotypic characterization of antimicrobial resistance in German Escherichia coli isolates from cattle, swine and poultry. Journal of Antimicrobial Chemotherapy, 52(3), 489–492.
- Lockley, A. K., & Bardsley, R. G. (2000). DNA-based methods for food authentication. Trends in Food Science & Technology, 11(2), 67–77.
- Martins, F. H., Guth, B. E. C., Piazza, R. M., Leao, S. C., Ludovico, A., Ludovico, M. S., Dahbi, G., Marzoa, J., Mora, A., & Blanco, J. (2015). Diversity of Shiga toxin-producing Escherichia coli in sheep flocks of Paraná State, southern Brazil. Veterinary Microbiology, 175(1), 150–156.
- Mullis, K., Faloona, F., Scharf, S., Saiki, R., Horn, G., & Erlich, H. (1992). Specific enzymatic amplification of DNA in vitro: the polymerase chain reaction. Biotechnology Series, 17.
- Purwaningsih, R., Lukita, B. L., & Ningsih, E. N. I. C. (2020). Detection of Salmonella typhimurium ATCC 14028 in supplement health product liquid preparation using Real-Time PCR (qPCR). Asian Journal of Natural Product Biochemistry, 18(2).
- Ribeiro Júnior, J. C., Tamanini, R., Soares, B. F., de Oliveira, A. M., Silva, F. G., da Silva, F. F., Augusto, N. A., & Beloti, V. (2016). Efficiency of boiling and four other methods for genomic DNA extraction of deteriorating spore-forming bacteria from milk. Semin. Cienc. Agrar. 37: 3069–3078.
- Sapiun, Z., Sophian, A., Abinawanto, M., Kamba, V., Damiti, S. A., & Luawo, H. (2020). Optimization of McFarland Turbidity Standards Value in Determining Template DNA as Reference in Salmonella typhimurium ATCC 14028 Test Using Real-Time PCR (qPCR). PalArch’s Journal of Archaeology of Egypt/Egyptology, 17(6), 10916–10922.
- Silva, G. A. da, Bernardi, T. L., Schaker, P. D. C., Menegotto, M., & Valente, P. (2012). Rapid yeast DNA extraction by boiling and freeze-thawing without using chemical reagents and DNA purification. Brazilian Archives of Biology and Technology, 55, 319–327.
- Sophian, A., Purwaningsih, R., Muindar, M., Igirisa, E. P. J., & Amirullah, M. L. (2021a). Detection of Salmonella typhimurium ATCC 14028 in Powder Prepared Traditional Medicines Using Real-Time PCR. Borneo Journal of Pharmacy, 4(3), 178–183.
- Sophian, A., Purwaningsih, R., Muindar, M., Igirisa, E. P. J., & Amirullah, M. L. (2021b). Use of Direct PCR Technique Without DNA Extraction in Confirmation Test for Salmonella typhimurium Bacteria on Meatball Samples. Borneo Journal of Pharmacy, 4(4), 324–332.
- Swa, Y. C. (2011). Successful Generation of DNA profiles using Direct PCR. Infrared Spectroscopic Technique for the Forensic Discrimination of Marker.
- Van Tongeren, S. P., Degener, J. E., & Harmsen, H. J. M. (2011). Comparison of three rapid and easy bacterial DNA extraction methods for use with quantitative real-time PCR. European Journal of Clinical Microbiology & Infectious Diseases, 30(9), 1053–1061.
- Wilson, I. G. (1997). Inhibition and facilitation of nucleic acid amplification. Applied and Environmental Microbiology, 63(10), 3741–3751.